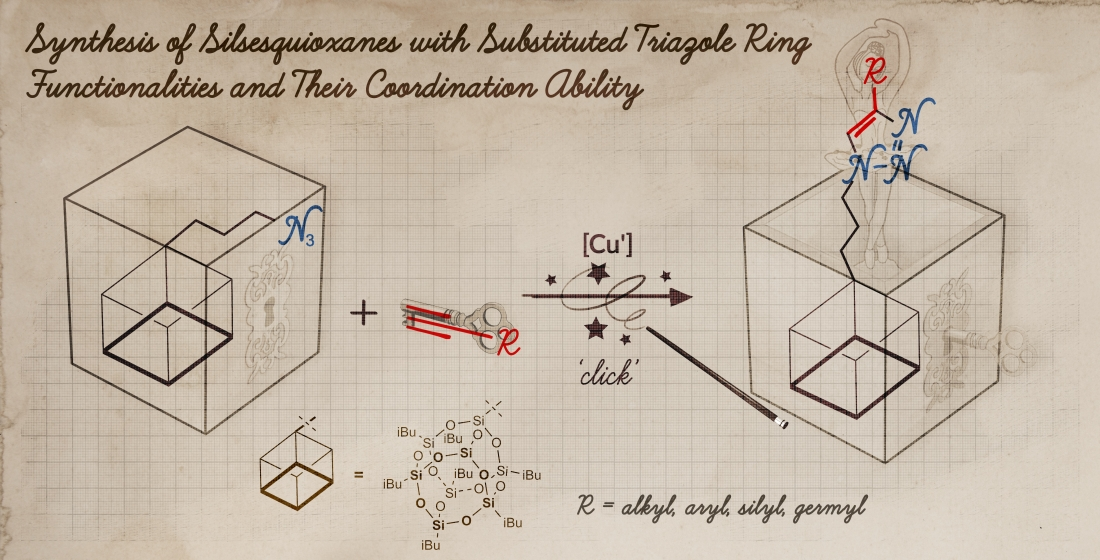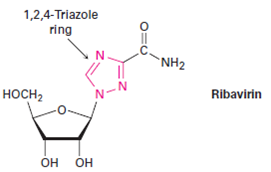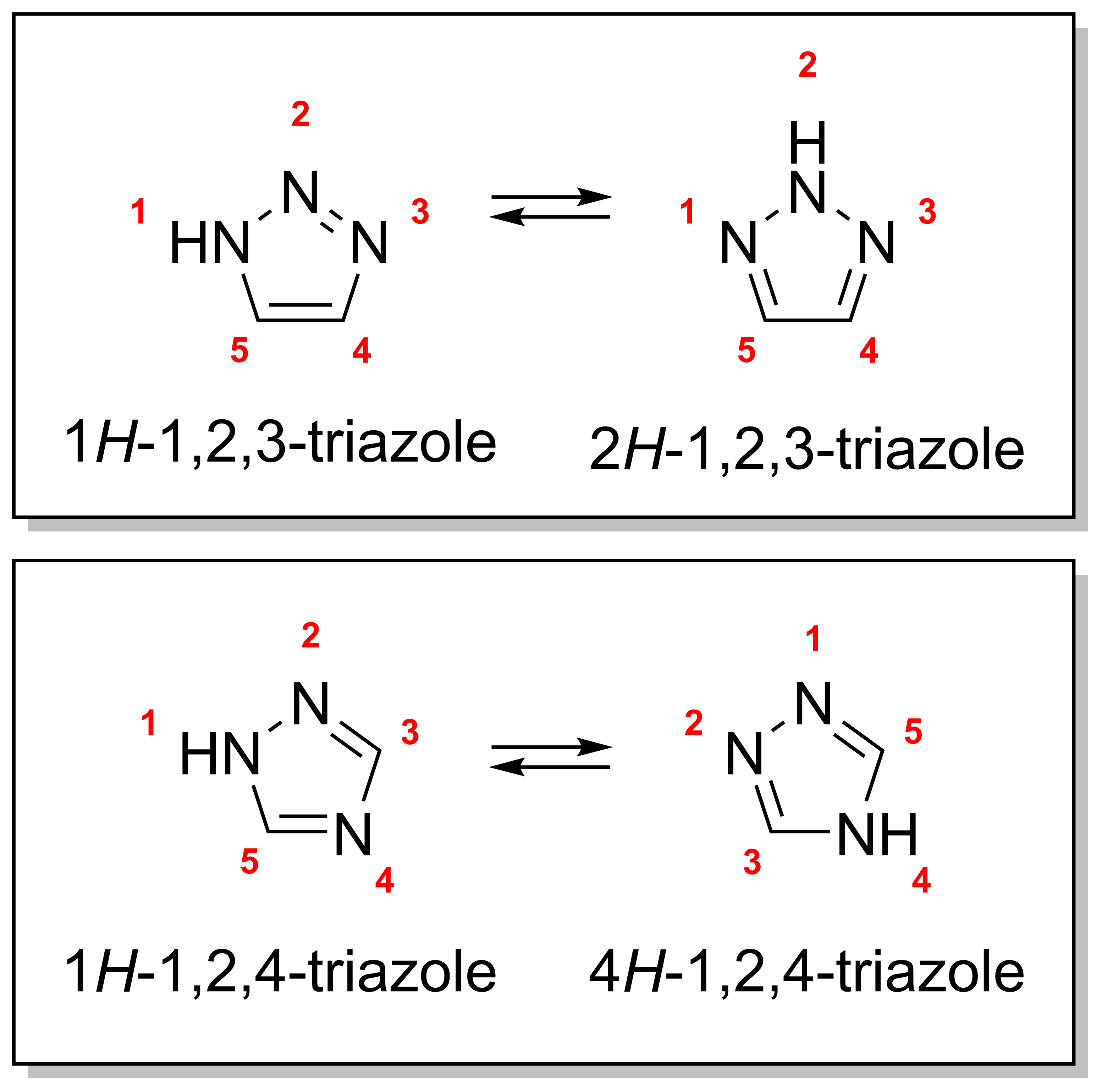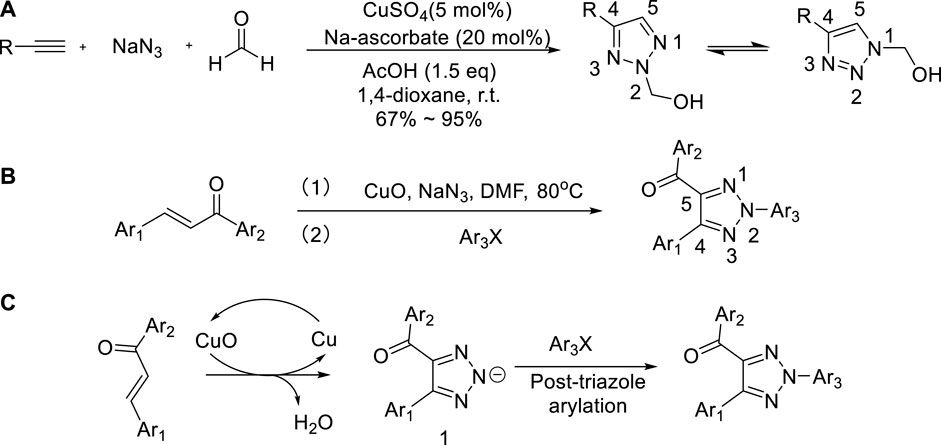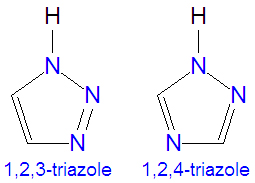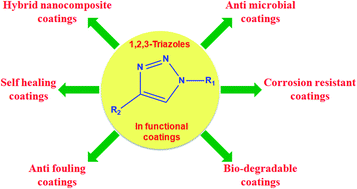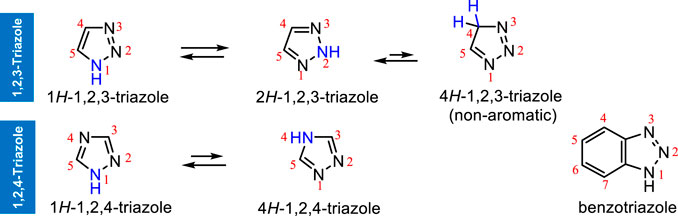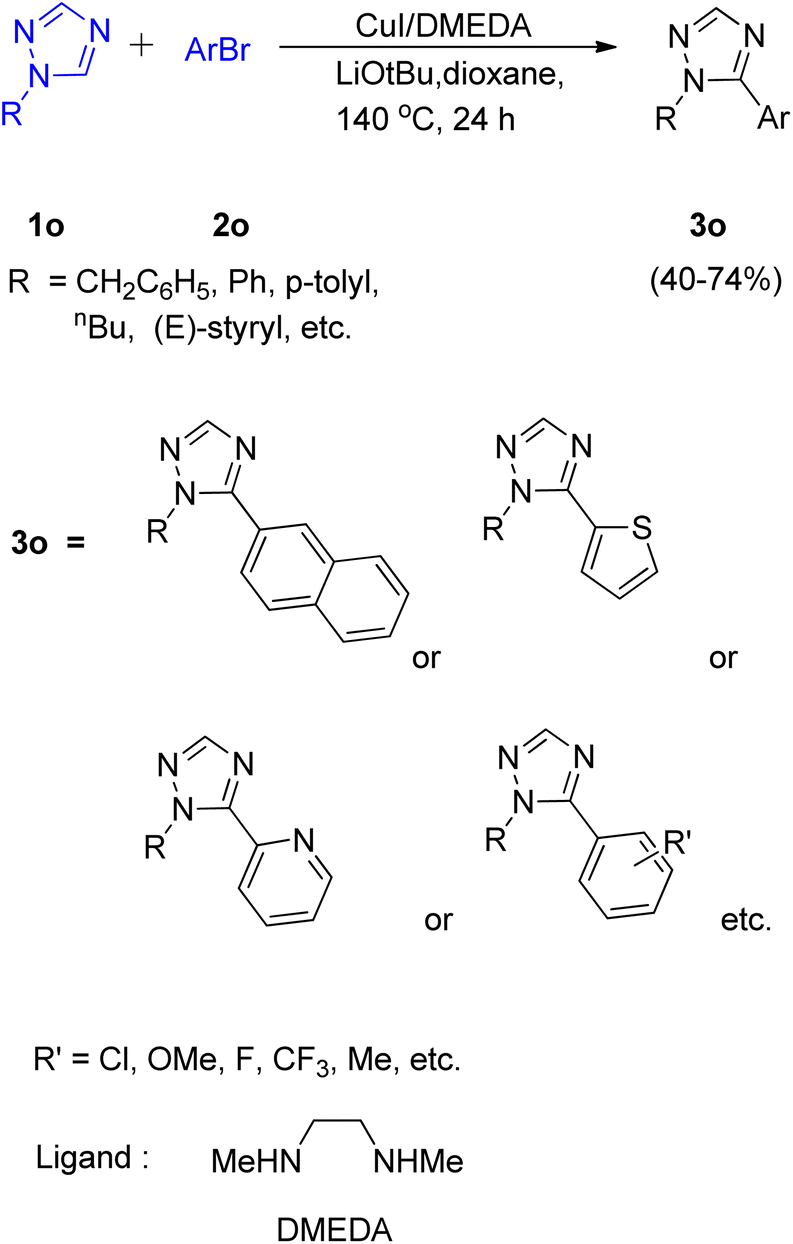
Metal catalyzed C–H functionalization on triazole rings - RSC Advances (RSC Publishing) DOI:10.1039/D2RA05697F
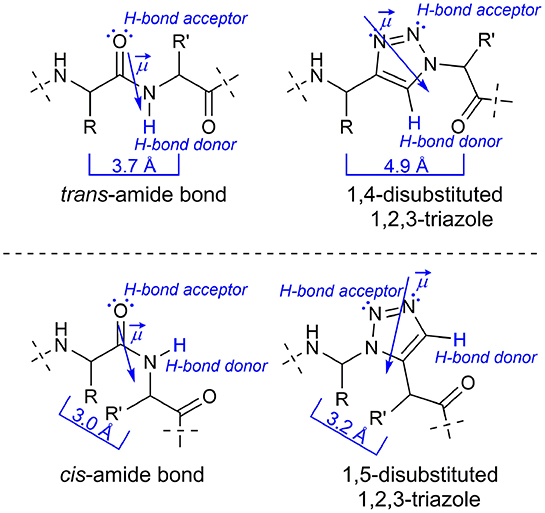
Frontiers | 1,4-Disubstituted 1H-1,2,3-Triazole Containing Peptidotriazolamers: A New Class of Peptidomimetics With Interesting Foldamer Properties

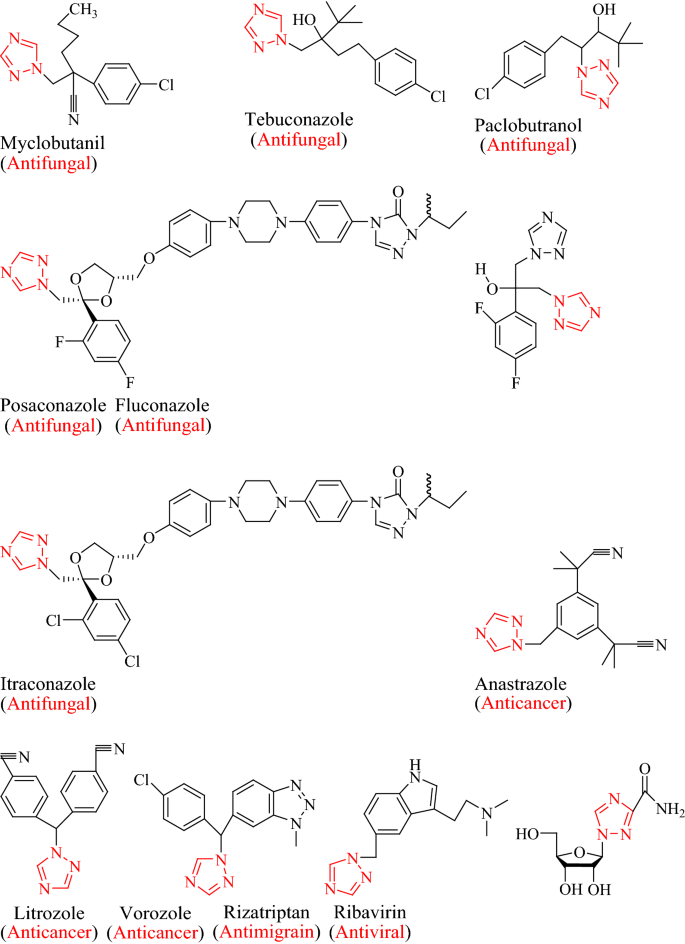
Click Chemistry: 1,2,3‐Triazoles as Pharmacophores - Agalave - 2011 - Chemistry – An Asian Journal - Wiley Online Library
Synthesis and biological evaluation of heterocyclic 1,2,4-triazole scaffolds as promising pharmacological agents | BMC Chemistry | Full Text

Thermochemistry and Initial Decomposition Pathways of Triazole Energetic Materials | The Journal of Physical Chemistry A
![PDF] Synthesis and Biological Evaluation of 1,2,3-Triazole Tethered Thymol-1,3,4-Oxadiazole Derivatives as Anticancer and Antimicrobial Agents | Semantic Scholar PDF] Synthesis and Biological Evaluation of 1,2,3-Triazole Tethered Thymol-1,3,4-Oxadiazole Derivatives as Anticancer and Antimicrobial Agents | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/af1a9a0c2811332b3ebbdaf37972b3ed3505467d/2-Figure1-1.png)
PDF] Synthesis and Biological Evaluation of 1,2,3-Triazole Tethered Thymol-1,3,4-Oxadiazole Derivatives as Anticancer and Antimicrobial Agents | Semantic Scholar

Synthesis of 1,4-Biphenyl-triazole Derivatives as Possible 17β-HSD1 Inhibitors: An in Silico Study | ACS Omega

Structural Consequences of the 1,2,3‐Triazole as an Amide Bioisostere in Analogues of the Cystic Fibrosis Drugs VX‐809 and VX‐770 - Doiron - 2020 - ChemMedChem - Wiley Online Library

A Recent Overview of 1,2,3-Triazole-Containing Hybrids as Novel Antifungal Agents: Focusing on Synthesis, Mechanism of Action, and Structure-Activity Relationship (SAR)

A Recent Overview of 1,2,3-Triazole-Containing Hybrids as Novel Antifungal Agents: Focusing on Synthesis, Mechanism of Action, and Structure-Activity Relationship (SAR)